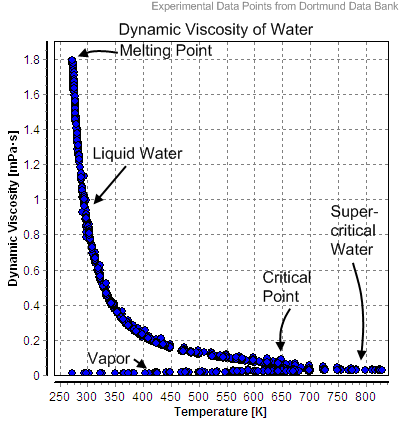
viscosity is how smoothly a liquid flows and how hardly the liquid flows it is highly viscous. The liquid with the most intermolecular force is high viscous. Is acetone more viscous than water Acetone has a viscosity 3x less than water but weighs 2.5 times more. So like its convenient density of 1 gm/cm 3, water at 20☌ provides a convenient comparison standard for viscosity. That is why, glycerol has a higher viscosity than water. That unit does not prove to be convenient for many uses, but note that the viscosity of water at 20☌ is almost exactly 1 if expressed in centipoise or milli-Pascal seconds. If you consider air and water, water has a higher viscosity than air as the air feels less resistance while flowing than water. For example, cooking oil, although rather inviscid at room temperature, becomes even less. For liquids such as water, viscosity can be perceived as a measure of the liquid’s resistance to flow. Higher concentration leads to a higher viscosity measurement. The more concentrated a polymer solution, the more large molecules you have exerting drag and interacting with each other. 
One of them is concentration, for example. Many liquids exhibit low viscosity at room temperature, such as water or alcohol, and other liquids demonstrate low viscosity under increasing heat. There are plenty of other factors you would have to take into account in using viscosity to assess molecular weight. This means that water molecules are more tightly packed together, resulting in higher. With the standard SI unit being the Pascal second or Poiseuille. Low viscosity in liquids is the result of temperature, with higher temperatures decreasing viscosity. Molecular weight: Water has a higher molecular weight than mercury. The viscosity of water increases significantly as temperatures decrease. Viscosity of liquids is expressed in a variety of units: Syrup is more viscous (has a higher viscosity) than water. The viscosity of water has been studied thoroughly because of the precision with which water flow must be controlled in many industrial applications. 
The low viscosity of water is important in its role as the solvent for life's chemical reactions and for blood circulation. The viscosity of water measures its resistance to flow and is critical to its many functions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
